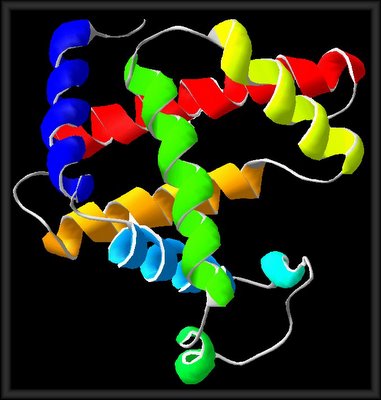
MOLECULAR BIOLOGY: ON HEDGEHOG PROTEINS: "TA "hedgehog protein" is a transmembrane protein involved in segment polarity and cell-cell signaling during embryogenesis and metamorphosis in the fruit fly Drosophila melanogaster, and in other insects and vertebrates. The expression and activity of Hedgehog proteins (Hh) exemplify a common strategy for pattern generation in metazoan embryos, namely, the specification of multiple cell fates through localized production and secretion of an instructive signal. In this manner, Hh signals regulate cell proliferation and differentiation in a diverse array of essential patterning events ranging from embryonic segmentation and appendage development in insects to neural tube differentiation in vertebrates
Study Sheds Light On Signaling Mechanism In Stem Cells, Cancer: "The primary cilium, it turns out, serves as the fulcrum in a series of acrobatic like moves between the Hedgehog signal and the Smoothened protein. Once Hedgehog has latched on to its receptor on the target cell's surface, it prompts the cell to move Smoothened, located in vesicles around the cell's nucleus, to the primary cilium. The positioning of Smoothened on the cilium, in turn, prompts downstream signaling of Hedgehog signals into the nucleus, where the instructions are issued. Just how or what the primary cilium is doing to promote Smoothened's activity is not clear, say the researchers." Hedgehog signals play an important role in prompting embryonic and adult stem cells to differentiate into some of the specialized cells that make up the body's tissues -- such as those of the brain, pancreas and skin. The scientists moved in on the role of Smoothened and the primary cilium incrementally. First, driven by their interest in Smoothened, they set out to determine where it was expressed in the embryo. They did so by developing highly specific antibodies to the protein and applying them to the tissue of an eight-day mouse embryo. The study revealed that Smoothened was modestly upregulated in cells of the node, an important early organizer tissue within the mouse embryo, and was expressed predominantly along the primary cilium of these nodal cells. This was a significant surprise. Second, to examine whether Smoothened's movement from vesicles around the nucleus to the cilium was regulated by Hedgehog signals, they carried out two studies, one involving cultured epithelial and fibroblasts cells expressing Smoothened, another involving a mouse embryo. In both cases, one set of cells was exposed to Hedgehog signals. Another set was exposed to cyclopamine, a drug that blocks Smoothened's function. In the cells exposed to the Hedgehog signals, Smoothened moved from the vesicles of the cell body to the cilium. In the cells exposed to cyclopamine, Smoothened was undetectable on the cilium. Scientists have known that cyclopamine inhibits Hedgehog signaling and can prevent Hedgehog-dependent cancers from spreading. The demonstration that the drug affected Smoothened movement to the cilium suggests how cyclopamine inhibits the Hedgehog pathway, the researchers say, and shows that the correlation between Smoothened on the cilium and pathway activation is very tight. Third, they examined whether the Smoothened protein included an amino acid sequence that other seven-transmembrane proteins require to move to the primary cilium and, if so, whether this sequence -- a so-called "motif" -- was essential to its relocation there. The answer to both questions was yes: A study of mouse cells in which Smoothened was mutated to lack the motif revealed that Smoothened no longer moved to the primary cilium. Finally, to determine Smoothened's function, they tested the mutant form of Smoothened that no longer could move to the primary cilium in epithelial cells in culture and in zebrafish embryos to see if the protein still functioned. It did not. "Thus, not only does Smoothened ciliary localization depend up on Hedgehog signaling, but Hedgehog signaling depends on a Smoothened ciliary localization motif," says Reiter. "Whether Smoothened functions at the cilium in all cell types remains to be determined. In addition, how Smoothened activates the Hedgehog pathway at the cilium remains unclear," he says. "But the current finding lays the groundwork for future studies that could ultimately have clinical benefit.""
The original news release can be found
here.